 
The atomic radius of an iron atom is about 156 picometers. Types of Radius with Respect to Types of Bonds. THE RADIUS OF AN ATOM DECREASES ALONG EACH PERIOD OF. 
Periodic Table of Elements - Sorted by Atomic Radius. How Big Are Atoms The atomic radius of the hydrogen atom is about 53 picometers. Atomic radius is generally stated as being the total distance from an atom’s nucleus to the outermost. THE ATOMIC RADIUS VARIES IN A PREDICTABLE AND EXPLICABLE WAY ACROSS THE PERIODIC TABLE AS SEEN PREVIOUSLY. 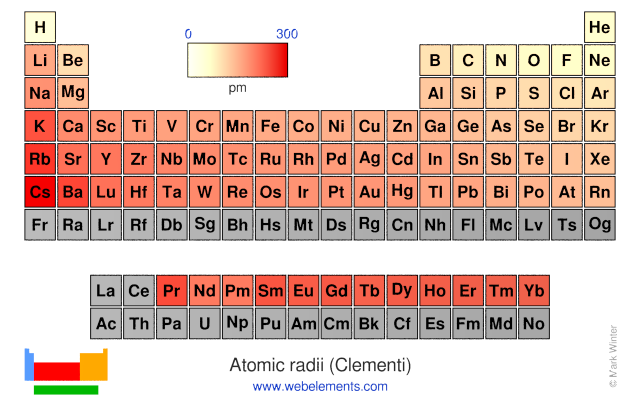
If you need to cite this page, you can copy this text: This increases the effective nuclear charge which pulls the electrons in closer towards the nucleus. Hence (ii) and (iii) are the correct representations. There is less shielding of these outer electrons. Explanation: Atomic radius is the distance between the nucleus and outermost shell consisting of electron (s). At the same time, the number of protons are increasing as we go across a period. But, the electrons we are adding are all in the same principal energy level and therefore not any farther away from the nucleus. This is curious since as we go across a period, we are adding electrons, just like we did going down a group. Increased nuclear charge, while the electrons that are added to the same principal energy level. Given Values- Atomic Radius (nm) 0.125 FCC Metal Gold BCC Metal: Vanadium Temperature (C) 1014 MetalA Barium Equilibrium Number of Vacancies (m3) 7. The electron distance from the nucleus determines the size of the atom.Īs we go across (left to right) a period on the periodic table, the atoms get smaller. Why?īecause as we go down a group, we have electrons in higher and higher energy levels which are farther away from the nucleus. Simply put, the atomic radius is half of the diameter of the atom, which is a result of the number of protons, neutrons, and electrons that compose the atom. Atomic Radius (radii) Trends and the Periodic TableĪs we go down any group on the periodic table, the atoms get larger. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
